Indication:
Dosage & Administration:
The recommended dosage in adults is a single loading dose of Remdesivir 200 mg (two vials of Remdinil™ 100 mg solution for IV infusion) on Day 1 followed by once-daily maintenance doses of Remdesivir 100 mg (one vial of Remdinil™ 100 mg solution for IV infusion) from Day 2 via IV infusion. For patients requiring invasive mechanical ventilation and/or ECMO, total treatment duration is 10 days. For patients not requiring invasive mechanical ventilation and/or ECMO, total treatment duration is 5 days. If a patient does not demonstrate clinical improvement, treatment may be extended for up to 5 additional days (i.e., up to a total of 10 days). Administer Remdesivir via IV infusion in a total volume of up to 250 mL 0.9% sodium chloride over 30 to 120 minutes.
Do not use Remdinil™ 100 mg solution for IV infusion for pediatric patients weighing 3.5 kg to less than 40 kg.
Preparation:
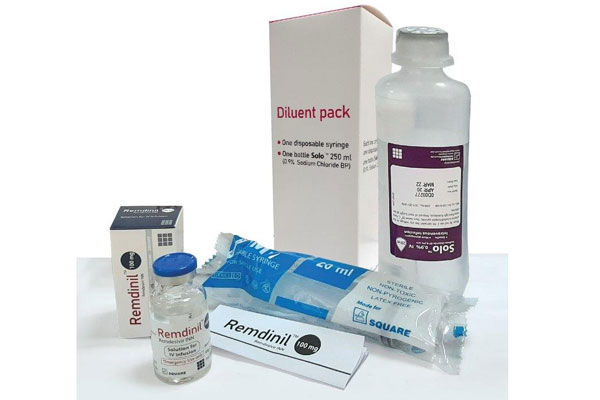
Remdinil™ 100 mg solution for IV infusion: 1 vial filled with Remdesivir 100 mg concentrate for solution for IV infusion, 20 ml disposable syringe & 250 ml 0.9% Sodium Chloride (Solo™) infusion bottle.